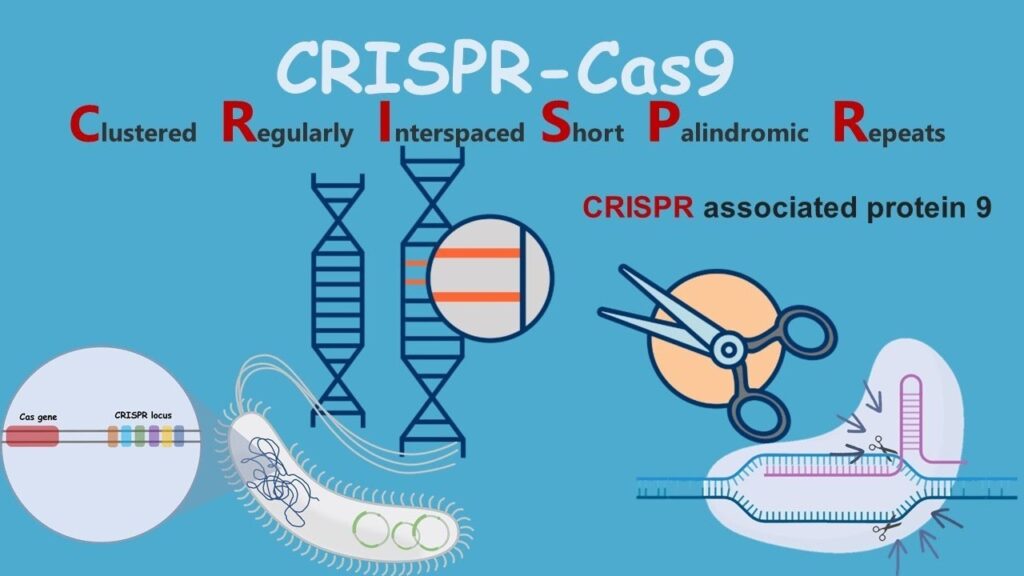
CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) technology has revolutionized the field of genetics, offering unprecedented precision and ease in gene editing. By allowing scientists to add, remove, or alter genetic material at specific locations within the genome, CRISPR has opened up vast possibilities for treating genetic disorders, advancing agricultural practices, and understanding fundamental biological processes. However, alongside its potential benefits, CRISPR also raises significant ethical and practical concerns. This blog explores the ethical and practical implications of CRISPR and gene editing, highlighting both its promise and the challenges it presents.
Understanding CRISPR Technology
CRISPR-Cas9 is the most well-known and widely used gene-editing system. It consists of two key components:
- The Cas9 Protein: An enzyme that acts like molecular scissors to cut DNA at a specific location.
- Guide RNA (gRNA): A piece of RNA that guides Cas9 to the precise spot in the genome where the cut is to be made.
Once the DNA is cut, the cell’s natural repair mechanisms can be harnessed to add or remove genetic material or to replace an existing DNA sequence with a new one.
Practical Applications of CRISPR
- Medical Therapies CRISPR has the potential to treat a wide range of genetic disorders. For example, researchers are exploring CRISPR-based treatments for conditions such as cystic fibrosis, muscular dystrophy, and sickle cell anemia. By correcting the genetic mutations responsible for these diseases, CRISPR could offer curative therapies where traditional treatments have failed.
- Cancer Research CRISPR is being used to develop new cancer treatments by targeting and modifying cancerous cells. It can help in identifying the roles of specific genes in cancer development, leading to more targeted and effective therapies.
- Agriculture In agriculture, CRISPR can be used to create crops that are more resistant to diseases, pests, and environmental stresses. It can also be used to enhance nutritional content, improve yield, and reduce the need for chemical inputs like pesticides and fertilizers.
- Basic Research CRISPR enables scientists to investigate the functions of specific genes with unprecedented precision. This can lead to new insights into genetic diseases, developmental biology, and the fundamental mechanisms of life.
Ethical Implications of CRISPR
- Germline Editing One of the most controversial aspects of CRISPR is its potential use in germline editing—modifying the genes in human sperm, eggs, or embryos. Changes made in the germline are heritable, meaning they can be passed on to future generations. This raises profound ethical questions about the potential for unintended consequences, genetic inequality, and the notion of “designer babies.”
- Equity and Access The benefits of CRISPR technology might not be equally accessible to all. There is a risk that only wealthy individuals or countries will be able to afford and benefit from these advanced treatments, exacerbating existing health and economic disparities.
- Informed Consent The complexity of gene-editing technologies makes it challenging to ensure truly informed consent. Patients and participants in gene-editing trials need to fully understand the potential risks, benefits, and uncertainties involved, which can be difficult to communicate effectively.
- Ecological Impact The use of CRISPR in agriculture and environmental management must be carefully regulated to prevent unintended ecological consequences. For instance, gene-edited organisms could potentially disrupt ecosystems if they were to spread unchecked in the wild.
- Regulatory and Governance Issues There is a need for robust regulatory frameworks to oversee the use of CRISPR technology. Currently, regulations vary widely between countries, and there is a lack of international consensus on many critical issues. Establishing clear guidelines and ethical standards is essential to prevent misuse and ensure that gene editing is conducted responsibly.
Practical Challenges of CRISPR
- Off-Target Effects One of the significant technical challenges with CRISPR is the potential for off-target effects—unintended cuts in the genome that can lead to unwanted mutations. While advancements are being made to improve the precision of CRISPR, ensuring its accuracy remains a critical area of research.
- Delivery Mechanisms Effectively delivering CRISPR components to the target cells in the body is another major challenge. Different tissues and cell types require different delivery methods, and finding safe and efficient ways to deliver CRISPR is essential for its success in clinical applications.
- Ethical Oversight Ensuring ethical oversight in research and clinical applications is paramount. Institutions and researchers must adhere to stringent ethical standards and guidelines to prevent abuses and ensure that the technology is used for the benefit of society.
- Long-Term Effects The long-term effects of gene editing are still largely unknown. Comprehensive long-term studies are needed to understand the potential risks and benefits fully and to monitor for any delayed adverse effects that might arise.
- Public Perception and Acceptance Public perception of CRISPR and gene editing plays a significant role in its acceptance and implementation. Misinformation and fear can hinder progress, so it is crucial to engage with the public, provide accurate information, and address concerns transparently.
CRISPR and gene editing hold immense promise for revolutionizing medicine, agriculture, and scientific research. However, their potential must be balanced with careful consideration of the ethical and practical implications. Addressing the challenges of off-target effects, delivery mechanisms, and long-term impacts, alongside robust ethical oversight and equitable access, is essential for harnessing the full potential of CRISPR responsibly. As the technology continues to advance, ongoing dialogue among scientists, ethicists, policymakers, and the public will be crucial to navigate the complex landscape of gene editing and to ensure that its benefits are realized in a just and sustainable manner.